Sulfuric Acid Treatment

GENERAL
Sulfuric acid is one of the world's most widely used chemicals and finds numerous applications as the production of fertilizers, explosives, plastic as e.g. polyurethanes, esterification of intermediates, dyes and also the iron and steel industries. The method used today for its production from sulfur respectively SO2 is the so called "Contact Process". SO2 is oxidized with oxygen on a heterogeneous catalyst - vanadium pentoxide contact - to SO3 and subsequent absorbed in ~98,5%wt sulfuric acid giving oleum which is rediluted with water down to 98.5%wt. Therefore this process typically produces sulfuric acid of 98.5% w/w concentration or stronger in the form of oleums.
Some of the processes and applications using sulfuric acid require weaker acid than this. As a result, dilution equipment is often necessary in order to meet the requirement for this weaker acid.
On the other hand in most chemical processes contamination and dilution of concentrated sulfuric acid occurs producing huge waste streams of contaminated spent sulfuric acid. An example is the production of Di-nitro-toluene where the waste acid of sulfuric acid is contaminated with nitric acid and partially nitrated toluene. Another example is the use of concentrated sulfuric acid for drying of gases like chlorine, bromine, hydrogen chloride or methyl chloride providing sulfuric acid with less than 80%w/w.
Recycling this spent sulfuric acid is either a precondition to run such processes profitably or generates considerable cost savings for raw material and effluent disposal. We build clean modern plants around the world to produce acid for re-use without contaminating the environment.
Here are some process examples you can take advantage of our extensive expertise and experience gathered during the past 50 years:
- Pre-concentration up to 70 wt%
- High-level concentration up to 98 wt%
- Chlorine drying with sulfuric acid incl. reconcentration of sulfuric acid
- Nitric acid concentration above the azeotropic point with sulfuric acid
- Hydrochloric acid concentration above the azeotropic point with sulfuric acid
- Denitration, concentration and recycling of sulfuric acid from e.g. various nitration processes
- Dilution of sulfuric acid
The System Sulfuric Acid / Water
For all of the above mentioned processes the behavior of the binary mixture sulfuric acid/water is important to know. The vapour liquid equilibrium data of sulfuric acid/water is shown below. It can be seen that up to a concentration of 70wt% sulphuric acid in the liquid phase almost no sulfuric acid is contained in the vapor phase. Above a concentration of 85wt% the vapor pressure of sulfuric acid is increasing significantly. At a concentration of slightly above 98,5%wt a maximum azeotop is formed at ambient pressure. Higher concentrations of sulfuric acid can only be realized by the addition of sulfurtrioxide or oleum.
Vapour/liquid Equilibrium Data of Sulfuric Acid/Water
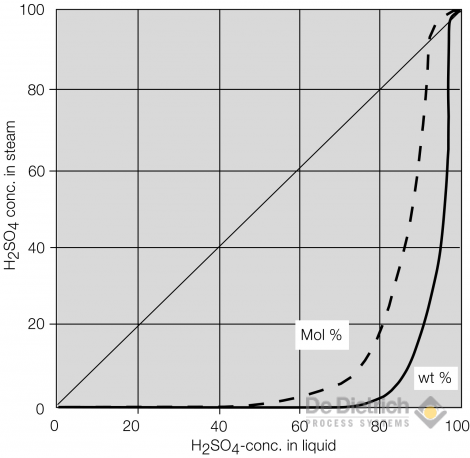
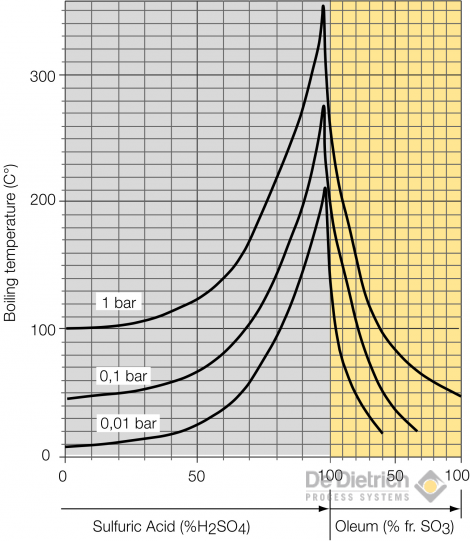
The following diagram shows that the boiling temperature at ambient pressure is quite high e.g. 338°C at the azeotropic point. Sulfuric acid at this high boiling temperature is very corrosive to almost any material of construction. Steam or oil at these temperatures are economically no more reasonable as heating media. The old fashioned route for re-concentrating sulfuric acid is therefore the so called pot or drum concentrator where the acid is either boiled with a burner in a silicon iron pot or injected with combustion gases into brick lined drums. The environmental consequence of these high temperature operations is the generation of exhaust fumes consisting of combustion products and often significant amounts of sulfuric acid mist.
Boiling temperature for 3 different pressures over the whole concentration range
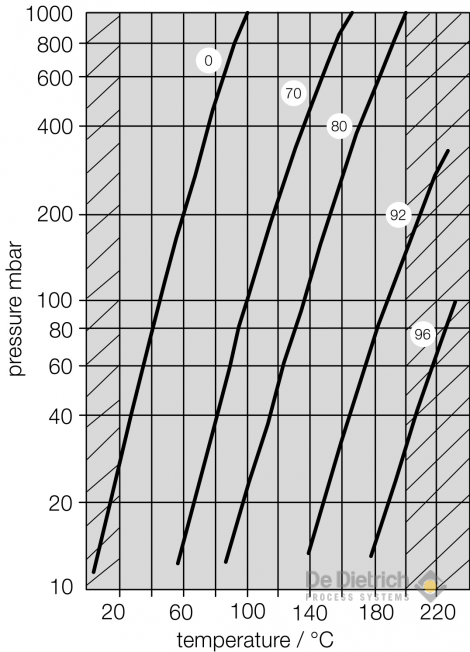
In order to overcome the environmental and corrosion problems we have developed a process to concentrate sulfuric acid under vacuum. This enables to concentrate sulfuric acid up to 98wt% at less than 210°C which permits the use of steam as heating media and the use of glass-lined steel, borosilicate glass 3.3, Tantalum and PTFE as material of construction. The lower limit of the operating pressure (vacuum) in the boiler is limited by the temperature of the cooling media which has to condense the evaporated water at this low pressure.
Vapour pressure as a function of the temperature for different sulfuric acid concentrations
.
Sulfuric Acid Recovery Plant

We provide highly efficient and environmental friendly process plants to recover, purify, concentrate or dilute sulfuric acid in a safe and reliabe manner over a wide concentration range even up to 98wt%.

